Key Takeaways
- MedCAD has received FDA 510(k) clearance for its AccuStride lower leg tibia and fibula system, a patient-specific surgical planning and guide tool.
- The system can speed up orthopedic surgeon workflows and provide customizable options for complex or revision lower leg region cases.
- MedCAD's solutions include vat-polymerized anatomical models, LPBF titanium guides, and implants for CMF, trauma, and cranial applications.
- The company's products are made in Texas, with a delivery time of approximately 5 days to US-based customers.
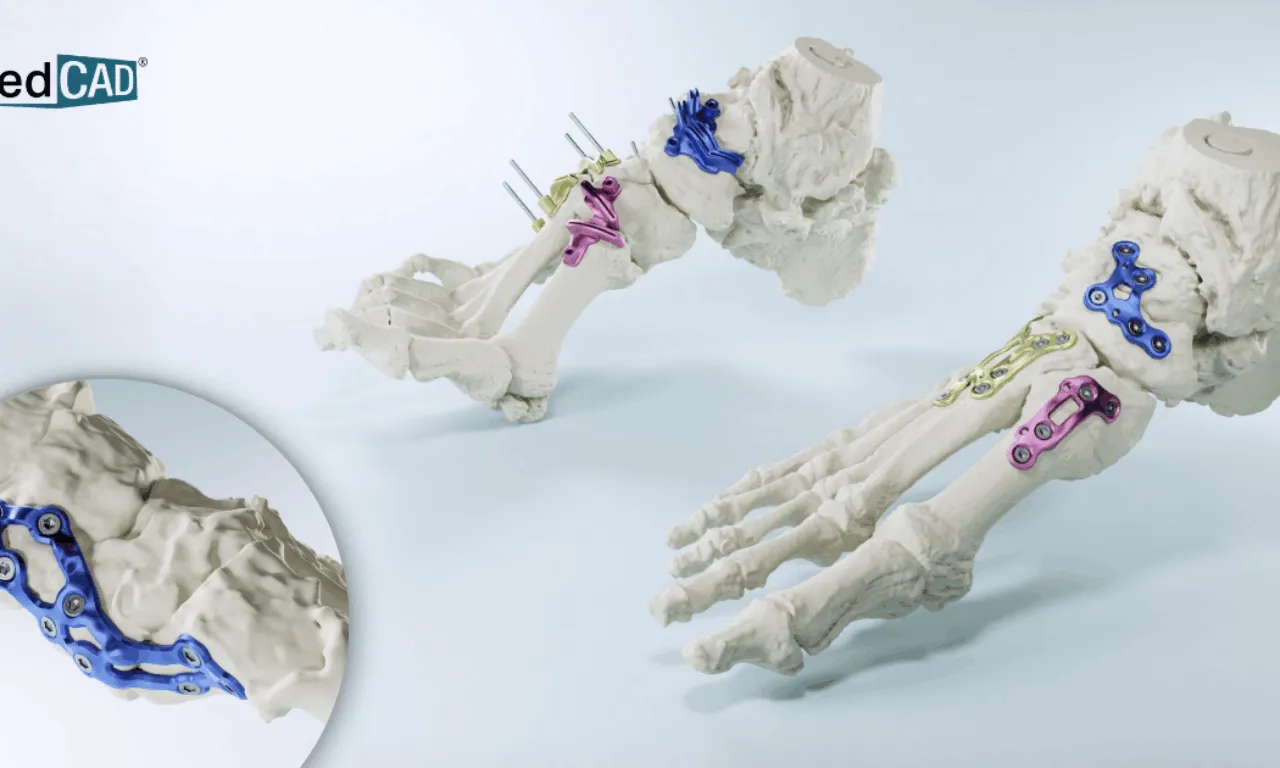
Introduction to MedCAD's AccuStride System
MedCAD, a leading provider of patient-specific surgical solutions, has announced the FDA clearance of its AccuStride lower leg tibia and fibula system. This innovative system is designed to streamline orthopedic surgeon workflows, particularly for complex or revision lower leg region cases. With this clearance, MedCAD can now offer a comprehensive solution, from CT scans to 3D printed guides, for feet, ankles, tibia, and fibula procedures.
Benefits of AccuStride
The AccuStride system provides several benefits, including:
- Patient-matched surgical guides that are anatomically unique to each patient
- Customizable options for multiple pathologies
- Potential to reduce the frequency and duration of surgeries
- Delivery of high-quality, durable outcomes
According to MedCAD CEO Nancy Hairston, "MedCAD's newly announced, patient-matched surgical guides are unlike anything else available for surgeons who routinely perform complex or revision lower leg region cases." The company's goal is to provide surgeons with cutting-edge planning technology to deliver life-changing outcomes for patients.
Comparison of MedCAD's Solutions
| Solution | Material | Application | Delivery Time |
|---|---|---|---|
| AccuStride | LPBF Titanium | Lower leg tibia and fibula | 5 days |
| Vat-Polymerized Anatomical Models | Photopolymer | Pre-surgical planning | 5 days |
| CMF Implants | Titanium | Craniofacial reconstruction | 5 days |
| Trauma Implants | Titanium | Fracture fixation | 5 days |
| Cranial Implants | Titanium | Neurosurgical procedures | 5 days |
Conclusion
MedCAD's AccuStride system has the potential to revolutionize orthopedic surgery by providing patient-specific, customizable solutions. With its FDA clearance, MedCAD can now offer a comprehensive range of products, from surgical guides to implants, for various orthopedic applications. The company's commitment to delivering high-quality, durable outcomes is evident in its use of advanced materials, such as LPBF titanium, and its focus on providing surgeons with cutting-edge planning technology.
Bottom Line
MedCAD's AccuStride system is a significant advancement in orthopedic surgery, offering patient-matched surgical guides and customizable options for complex or revision lower leg region cases. With its FDA clearance and comprehensive range of products, MedCAD is poised to make a significant impact in the medical industry, providing surgeons with the tools they need to deliver life-changing outcomes for patients.